Radioactive Materials
Home > Radiation Protection and Quality Assurance > Radiation Physics and Biology > Radioactive Material
Can we please get your advice on this one question?
Radioactive materials consist of materials, usually industrial wastes or by-products enriched with radioactive elements found in the environment. A radioactive element is an element with an unstable nucleus, which radiates alpha, beta or gamma radiation and gets converted to a stable element in a process called radioactivity. These unstable nuclei are called radionuclides and their emissions are referred to as ionizing radiation.
There are two types of radioactive materials:
- Naturally Occurring Radioactive Materials (NORM) and
- Technologically Enhanced Naturally Occurring Radioactive Materials (TENORM)
Naturally-occurring radioactive materials (NORM) exist in nature and are brought to the surface through human activities such as oil and gas exploration or mining. Natural radioactivity is the decay process of the naturally occurring elements, usually heavy elements, that are heavier than lead. Natural radioactive atoms heavier than lead cannot attain a stable nucleus heavier than lead. The natural background exposures are frequently used as a standard of comparison for exposures to various artificial sources of ionizing radiation.
Artificial radioactive atoms are produced either as a by-product of fission of uranium or plutonium atoms in a nuclear reactor or by bombarding atoms with particles (such as neutrons, protons, or heavy nuclei) at high velocity via a particle accelerator. The produced radioactive elements decay by emitting particles, such as alpha particles, positive or negative beta particles, and energy photons (gamma rays).
Radioactive materials are used in medicine in diagnostic and therapeutic procedures. Diagnostic procedures using radioactive materials, such as those used in nuclear medicine, involve the use of relatively small amounts of radioactive materials to facilitate imaging of certain organs. Therapeutic uses of radioactive materials include teletherapy, brachytherapy, and therapeutic nuclear medicine to kill cancerous tissue, reduce the size of a tumor or reduce pain.
Beam Characteristics
Radiation is scattered and absorbed when it passes through tissue. The intensity of the radiation attenuates exponentially within the absorbent thickness (tissue), using the following exponential law of absorption:
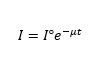
Where;
I = Intensity of the outgoing radiation beam
I° =Intensity of the incoming radiation beam
µ = absorption coefficient
t = tissue thickness.
The intensity of the outgoing radiation depends on the tissue absorption coefficient and its thickness.
Types of interactions of radiation with matter
1) Photoelectric Effect
Also known as the Hertz effect was first observed by Heinrich Rudolf Hertz in 1887 before being published by Albert Einstein in 1905. It was defined to as the emission of electrons or free carriers when light shines on a material.
When electromagnetic radiation reaches a surface (generally a metallic surface), it transfers its energy to the electrons of the surface and on reaching or exceeding the threshold frequency, they propel from the inner atomic orbital or get scattered.
It is dominant at energies approaching zero or less than 35kV and with high atomic number (z). It is applied in diagnostic radiology because the atomic number of bone is higher than that of soft tissue; hence the absorption of the incoming radiation is higher in bone than in soft tissue.
1) Compton effect
It was discovered in 1923 by Arthur Holly Compton, it is the interaction of the x-ray or gamma photon with free electrons or loosely bound valence shell electrons. The resultant incident photon gets scattered and imparts energy to the electrons of other atoms. The scattered photon will have a longer wavelength and a lower energy.
It is dominant at
energies of between 35Kv to 50MV. It has no dependency on the atomic number (Z) but it depends on the
electron density of the material. It is applied in radiotherapy in the
absorption of ionizing radiation because of the uniformity of the absorption of
the incoming radiation both in bone and in soft tissue.
2) Pair Production
Pair production refers to a photon creating an electron-positron pair near a nucleus. The electron sheds all of its energy by the absorption processes while the positron propagates through the medium ionizing atoms until its energy has drops to a lower level that it pulls a free electron close enough to combine with it. For pair production to occur, the incoming energy of the interaction must be above the threshold photon energy level of 1.02MeV.
It is dominant at higher energies of more than 10MeV and it increases with increase in square of the atomic number of the nearby atom.
When the nucleus of a radioactive nuclide spontaneously gives up its extra energy, this energy is known as ionizing radiation. Ionizing radiation may take the form of alpha particles, beta particles, or gamma rays, or x-rays. The phenomenon of emitting the radiation is called radioactive decay. The sources of radioactive materials
Nuclear reactors:
- Radiotherapy installations using the radioisotope machines
- Nuclear medicine department in a hospital
- Research institutes using the radioisotopes
- Inherent radiation sources in the earth crust.
All of the above mentioned sources are affecting the environment in certain way; the ways will briefly discussed below.
Nuclear Reactors:
Nuclear reactors using the radioactive materials such as uranium, thorium, plutonium for generating the power production r purposes, nuclear bomb production and other research purposes. When they are using like this plenty of advantages are there however, that much of hazards is also there. If, they are not handled or disposed in secured places this things will affect the environment in potential way. In the Second World War shows the effect of radiation to this world through the explosion of atom bomb. These things will also lead to an environmental hazard if it happens again for several years.
Radiotherapy and Nuclear Medicine Department in a Hospital:
Radiotherapy and Nuclear medicine department are playing a vital role in cancer treatment and its diagnosis. For the treatment purposes radioisotopes are used in general in developing countries in addition to that linear accelerators also in use. Because of this greater use of the radioactive it should special attention is required in maintaining the import, export, and disposal of these sources. In all the country the respective government body is taking the steps to monitor that. However, these sources are causing some environmental effect when they are using for treatment.
Inherent Radiation Sources in Earth Crust:
The inherent radiation sources in earth crust are the other major factor in causing the environmental pollution. Because the radiation is level is different for different parts of the world. Especially Kerala coastal an area in India is having the natural uranium sources this place is having the natural background radiation than other parts of the country.
The environmental protection can only stabilized or bring to the normal position or in control by reduction in the usage of radioactive materials in day today life in the world.
References:
- http://www.nj.gov Beyzadeoglu M.; Ozyigit G.; Ebruli. (2010). Basic Radiation Oncology. Retrieved 2018-13-08
- NCBI. (2013 Feb). Overview of Basic Radiation Physics, Chemistry and Biology. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK158810/
- U.S.NRC. (2011, Feb 04). Backgrounder on Medical Use of Radioactive Materials. Retrieved from https://www.nrc.gov/reading-rm/doc-collections/fact-sheets/med-use-radioisotopes-bg.html
- Wikipedia Encyclopedia. (2018, August 8). Compton scattering. Retrieved from https://en.wikipedia.org/wiki/Compton_scattering
- Wikipedia Encyclopedia. (2018, April 24). Naturally occurring radioactive material. Retrieved from https://en.wikipedia.org/wiki/Naturally_occurring_radioactive_material
- Wikipedia Encyclopedia. (2018, July 13). Pair production. Retrieved from https://en.wikipedia.org/wiki/Pair_production
- Wikipedia Encyclopedia. (2018, August 9). Photoelectric effect. Retrieved from https://en.wikipedia.org/wiki/Photoelectric_effect
Home > Radiation Protection and Quality Assurance > Radiation Physics and Biology > Radioactive Material
FREE Infographic What successful people believe. What successful people do
Dictionary of Cancer Terms
Need help understanding a word? Here is an electronic resource that gives meaning to Cancer terms and their usage.

StrengthsFinder 2.0