Radiation
Energy
Home > Treatment Planning > Prescription & Dose Calculation > Treatment Prescription > Radiation Energy
Can we please get your advice on this one question?
Definition:
Energy is the capacity of a physical system to perform work. Energy exists in several forms such as heat, kinetic or mechanical energy, light, potential energy, electrical, or other forms.
The SI unit of energy is the joule (J) or Newton-meter (N * m).
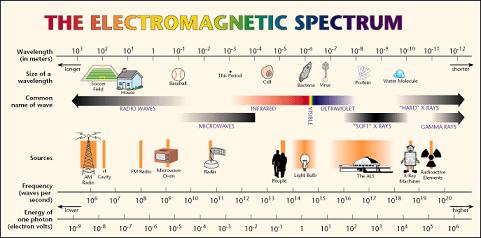
Figure (a) the electromagnetic spectrum is the distribution of electromagnetic radiation according to energy.
Each and every object has some energy on own, starting from the radio waves, man, x-ray machines and radioactive sources. Our area of interest is on the high energy x-ray and radioactive sources which emits high energy as well as low energies to treat the cancer.
Selection of energy for the treatment of cancer is the most crucial factor. If there is any mistake happens the tumour could be underdosage or overdosage. To avoid such circumstances selection of energy must be given particular attention for each patient during the treatment planning. Many types of radiation are used in the treatment they are,
1. X-rays or gamma rays
2. Alpha particles
3. Electron or beta particles
4. Proton therapy
5. Neutron etc.,
Each one said above has different type of interaction mode when it interacts with the matter so that, energy distribution also be vary.
As mentioned previously, the energy of the radiation is responsible for its ability to penetrate matter. Higher energy radiation can penetrate more and higher density matter than low energy radiation. The energy of ionizing radiation is measured in electronvolts (eV). One electronvolt is an extremely small amount of energy so it is common to use kiloelectronvolts (keV) and megaelectronvolt (MeV). An electronvolt is a measure of energy, which is different from a volt which is a measure of the electrical potential between two positions. Specifically, an electronvolt is the kinetic energy gained by an electron passing through a potential difference of one volt. X-ray generators have a control to adjust the keV or the kV.
The energy of a radioisotope is a characteristic of the atomic structure of the material. Consider, for example, Iridium-192 and Cobalt-60, which are two of the more common industrial Gamma ray sources. These isotopes emit radiation in two or three discreet wavelengths. Cobalt-60 will emit 1.33 and 1.17 MeV Gamma rays, and Iridium-192 will emit 0.31, 0.47, and 0.60 MeV Gamma rays. It can be seen from these values that the energy of radiation coming from Co-60 is about twice the energy of the radiation coming from the Ir-192. From a radiation safety point of view, this difference in energy is important because the Co-60 has more material penetrating power and, therefore, is more dangerous and requires more shielding.
Question:
1. Cobalt-60 will emits radiation of energy,
a) 1.33 and 1.17 MeV
b) 1.33 and 1.18 Mev
c) 1.30 and 1.20 Mev
d) 1.30 and 1.17 Mev
Answer
1. a) 1.33 and 1.17 Mev
References:
Home > Treatment Planning > Prescription & Dose Calculation > Treatment Prescription > Radiation Energy
FREE Infographic What successful people believe. What successful people do
Dictionary of Cancer Terms
Need help understanding a word? Here is an electronic resource that gives meaning to Cancer terms and their usage.

StrengthsFinder 2.0
